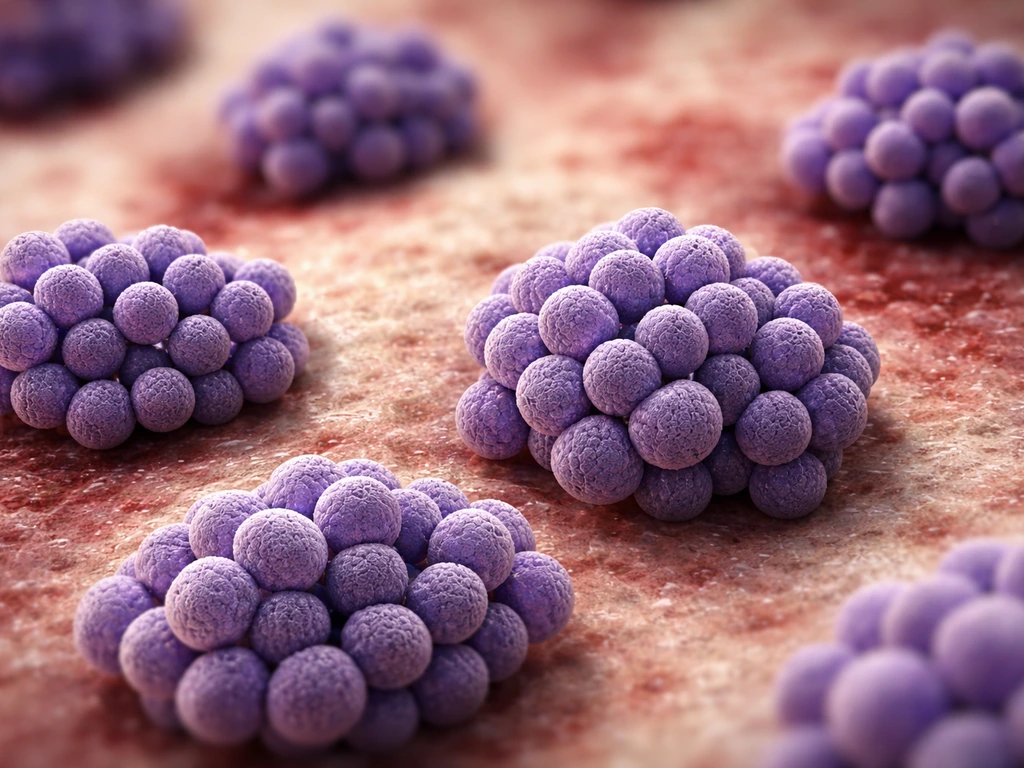
Pus-forming bacteria that grow in bunches or clusters are almost always staphylococci, and the one you need to know first is Staphylococcus aureus. When a microbiologist looks at an infected wound sample under the microscope after a Gram stain, those irregular, grape-like clusters of purple (Gram-positive) round cells are a strong visual signal that you are dealing with a staphylococcal species. This article explains exactly what that means, which organisms are involved, how labs confirm them, what conditions let them grow, and what to do about it practically.
Pus-Forming Bacteria in Clusters: How to Identify and Control Them
What 'pus-forming' and 'clusters' actually mean
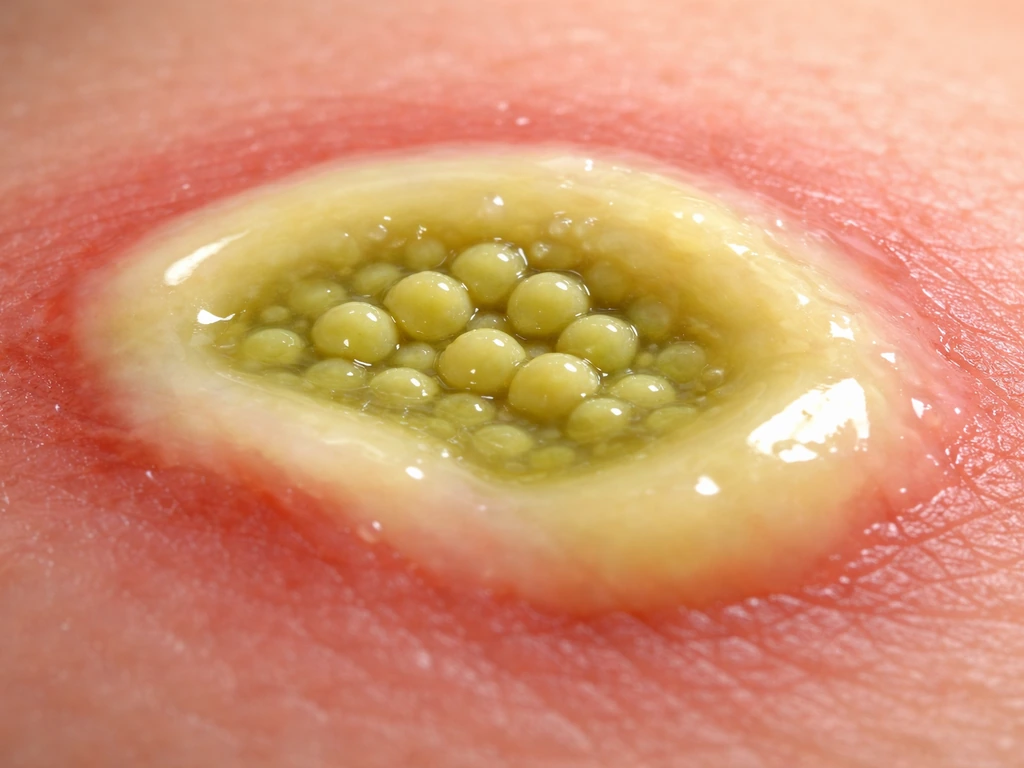
Pus is the yellowish or greenish fluid that accumulates at an infection site. It is made up of dead white blood cells (neutrophils), tissue debris, and live or dead bacteria. Bacteria that consistently trigger this kind of intense neutrophil response are called pyogenic, from the Greek for "pus-producing." Not every bacterium does this as reliably or dramatically as staphylococci do.
The "clusters" or "bunches" description comes directly from how these bacteria reproduce. Staphylococci divide along multiple axes, meaning each daughter cell can split in a different direction. The result under the microscope is an irregular, grape-like cluster rather than a tidy line. Streptococci, by contrast, divide along a single axis and line up into pairs or chains. That one visual difference is one of the first things a lab technician uses to narrow down which organism they are looking at.
The main culprits: Staphylococcus species
Staphylococci are pus-forming bacteria that grow in clusters, and the genus contains many species, but a few come up again and again in clinical and food-safety contexts. Here is a quick breakdown of the most important ones.
Staphylococcus aureus
S. aureus is the headline organism for this category. It is coagulase-positive, meaning it produces an enzyme that clots plasma, which is a key lab marker. Its virulence toolkit is extensive: it produces leukotoxins, hemolysins, and other toxin families that help it evade white blood cells and destroy tissue. This is why S. aureus infections tend to produce the most dramatic abscesses and the most aggressive spread. It is also the organism behind the MRSA problem discussed later.
Coagulase-negative staphylococci (CoNS)
This group includes species like Staphylococcus epidermidis and Staphylococcus saprophyticus. They are normal residents of human skin and mucous membranes, which sounds harmless until you consider the situations where they become a problem. S. epidermidis is a leading cause of infections on implanted devices, catheters, and prosthetic joints, largely because it forms tenacious biofilms on foreign surfaces. S. saprophyticus is a well-recognized cause of urinary tract infections, particularly in young, sexually active women, and can also be involved in more serious complications like pyelonephritis. These organisms are less virulent than S. aureus individually, but their ability to form biofilms makes them genuinely difficult to eradicate.
How cluster-forming cocci differ from chain-formers
It is worth being clear about what cluster-forming staphylococci are not. Streptococci, including species like S. pyogenes (Group A Strep) and S. pneumoniae, are also Gram-positive cocci and also cause purulent infections. But they form pairs or chains under the microscope, not clusters, and they test catalase-negative where staphylococci test catalase-positive. If someone tells you they saw "round bacteria in clusters" on a Gram stain, that points to Staphylococcus. Chains point elsewhere. Getting this right early matters because treatment choices can differ.
How labs confirm cluster-forming pus bacteria

Lab confirmation follows a logical sequence, and understanding each step helps you interpret a lab report or know what to expect when a sample is sent for culture.
- Gram stain: The first and fastest step. A smear of the sample is stained and examined under the microscope. Gram-positive cocci in irregular clusters immediately suggest Staphylococcus. This result alone guides the initial clinical response while culture results are pending.
- Catalase test: A drop of hydrogen peroxide is placed on the colony. Staphylococci produce catalase, so they bubble (oxygen is released). Streptococci do not. This one simple test separates the two major groups of Gram-positive cocci.
- Mannitol salt agar (MSA): This is a selective and differential medium. It contains 7.5% NaCl, which suppresses most organisms but not staphylococci. S. aureus ferments mannitol and turns the agar yellow; coagulase-negative species typically do not ferment mannitol and leave the agar pink or red. MSA combined with DNase testing improves the accuracy of identification significantly.
- Coagulase test: The definitive test for separating S. aureus from CoNS. The slide coagulase test involves emulsifying a colony in a drop of undiluted citrated plasma and reading for visible coarse clumping within about 10 seconds. The tube coagulase test detects free coagulase using diluted plasma and is read over several hours. A positive result confirms S. aureus.
- Rapid confirmatory methods: Latex agglutination kits and testing for protein A, clumping factor, and thermostable nuclease are used as confirmatory steps in many lab workflows. MALDI-TOF mass spectrometry has also become a reliable rapid identification tool for Staphylococcus species in routine clinical labs.
Growth conditions: what these bacteria need to thrive
This is where the food-safety and contamination-prevention angle gets practical. S. aureus is unusually tough compared to many foodborne pathogens. Knowing its growth thresholds tells you exactly what environmental controls you need to apply.
| Condition | Growth Range | Optimum | Practical Note |
|---|---|---|---|
| Temperature | ~7°C to 47.8°C (44.6°F to 118°F) | 35–37°C (95–98.6°F) | Grows at refrigerator-borderline temps; freezing stops growth but doesn't kill it |
| pH | ~4.0 to 9.8 | 6.0–7.0 | Survives mild acid/base conditions that would inhibit many other pathogens |
| Water activity (a_w) | ~0.86–0.90 minimum (aerobic vs anaerobic) | >0.99 | Can grow in salt-cured and dried foods where other pathogens cannot |
| Salt (NaCl) tolerance | Up to ~10–20% | Low to moderate | More salt-tolerant than most foodborne bacteria; survives in brined products |
| Oxygen requirement | Facultative anaerobe | Aerobic preferred | Grows in wounds, abscesses, and low-oxygen food packaging environments |
The facultative anaerobe status is particularly important. S. aureus can grow both aerobically and anaerobically, which explains why it thrives in sealed abscess cavities, deep wounds, and modified-atmosphere food packaging. Understanding where biofilms grow helps put this in context, because staphylococcal biofilms on surfaces and devices form in exactly these kinds of low-oxygen, nutrient-rich microenvironments.
The low minimum water activity (down to about 0.86 under certain conditions) is a big deal for food safety. Most bacteria cannot grow below about 0.91. S. aureus can, which means it can colonize salty, dried, or partially dehydrated foods that seem too inhospitable for contamination. This is why hand hygiene matters so much in food preparation, not just in clinical settings.
What these bacteria can actually do to you
The range of conditions staphylococci cause is wider than most people realize. Here is a summary of the main clinical presentations to watch for.
- Skin and soft-tissue infections: Boils, carbuncles, folliculitis, and abscesses are the most common presentations. The abscess is the classic sign, a painful, fluctuant, pus-filled lump, often with surrounding redness and warmth.
- Wound infections: Surgical wounds, cuts, and scrapes can all become colonized. Purulent drainage from a wound is the key sign.
- Food poisoning: S. aureus produces heat-stable enterotoxins in contaminated food. Symptoms (nausea, vomiting, cramping, diarrhea) typically begin 1–6 hours after eating the contaminated food and resolve within 24–48 hours. The toxin, not the live bacteria, causes the illness.
- Urinary tract infections: Primarily S. saprophyticus in young women; presents with dysuria and urinary frequency.
- Device-related infections: S. epidermidis and other CoNS on catheters, prosthetic valves, and joint replacements.
- Bacteremia and sepsis: Fever combined with low blood pressure can signal that bacteria have entered the bloodstream. This is a medical emergency.
Knowing when to escalate is critical. The IDSA defines systemic inflammatory response (SIRS) criteria that signal more serious infection: temperature above 38°C or below 36°C, breathing rate above 24 breaths per minute, heart rate above 90 beats per minute, and white blood cell count above 12,000 or below 400 cells per microliter. If any of those signs appear alongside a skin infection, that changes the management picture immediately.
For day-to-day wound monitoring, the CDC's guidance on cellulitis is a useful rule of thumb: seek medical attention right away if the red area around a wound spreads quickly or if you develop fever or chills. Spreading redness plus fever together is the combination that should prompt same-day evaluation, not a wait-and-see approach.
Treatment and the MRSA problem
For a simple abscess, incision and drainage is still the primary treatment. Physically opening and draining the pus removes the bacterial load directly and, for uncomplicated cases in healthy individuals, that may be sufficient without antibiotics. The IDSA is clear on this point: antibiotics are an adjunct in certain circumstances, not the automatic first step for every abscess.
Antibiotics are added when there are signs of systemic involvement (the SIRS criteria above), when the infection is spreading, or when the patient has risk factors for complicated infection. For recurrent abscesses, the approach shifts: culture the abscess first, then treat with an antibiotic active against the specific pathogen for 5 to 10 days, adjusting based on clinical response.
MRSA (methicillin-resistant Staphylococcus aureus) changes the antibiotic selection conversation significantly. MRSA strains are resistant to most penicillin-class antibiotics and many related drugs. This is not a rare hospital curiosity anymore. Community-acquired MRSA infections show up in otherwise healthy people with no healthcare exposure. The pus from an infected wound can contain MRSA, and standard beta-lactam antibiotics will not clear it. Labs that confirm MRSA will guide clinicians toward alternative agents such as clindamycin, trimethoprim-sulfamethoxazole, or in serious cases, intravenous vancomycin.
The antibiotic resistance angle also ties into the broader history of how treatments were developed. For context, it is interesting to note that the story of where penicillin grows naturally begins with a Staphylococcus contamination event in Alexander Fleming's lab, and the question of does penicillin grow on bread gets at the same mold biology that gave us our first antibiotic against these very organisms. The fact that staphylococci have since evolved resistance to so many penicillin-class drugs is a direct result of decades of selective pressure.
Prevention: what you can actually do today
Wound and personal hygiene
Keeping cuts and scrapes clean and covered with a bandage or dressing is the most straightforward step. Change dressings with clean hands, and wash hands before and after. Do not share personal items like towels or razors, because staphylococci transfer easily on skin-contact surfaces. If a wound is draining pus, treat that drainage as potentially containing MRSA and handle it accordingly with gloves and proper disposal.
Food safety and preventing staphylococcal food poisoning

S. aureus food poisoning is almost entirely preventable with temperature control. The bacteria need time at warm temperatures to multiply enough to produce dangerous toxin levels. The CDC and USDA both recommend refrigerating perishable foods within 2 hours of preparation or purchase, or within 1 hour if the ambient temperature is 90°F (32°C) or above. This is not just a general guideline; it is the specific window within which S. aureus can start multiplying toward toxin-producing concentrations.
Cross-contamination from hands to food is the main transfer route in kitchen settings. Handwashing before food handling, particularly after touching wounds or skin, is essential. Someone with an active skin infection should not be handling food that will not be cooked further. Understanding what round bacteria that grow in clusters are called in a microbiology context helps food safety professionals make the connection between the Gram stain result and the specific contamination risk they are managing on the production floor.
Surface and environmental control
In healthcare settings, environmental cleaning and barrier precautions are the primary tools for reducing MRSA transmission. In food production, regular sanitation of contact surfaces, monitoring of temperature logs, and handwashing compliance are the equivalent controls. S. aureus is not particularly resistant to standard disinfectants when surfaces are properly cleaned, but it is very good at persisting on dry surfaces and reforming contamination after inadequate cleaning.
One more layer worth knowing: staphylococci are experts at forming biofilms on both biological and non-biological surfaces. A biofilm is a community of bacteria encased in a protective matrix that makes them much harder to kill with either antibiotics or disinfectants than free-floating cells. Once a biofilm establishes on a food-contact surface or a medical device, routine cleaning concentrations may not be enough to remove it. This is why consistent, thorough cleaning protocols matter more than occasional intensive ones.
FAQ
If a Gram stain shows grape-like Gram-positive clusters, does that automatically mean MRSA or just staphylococci?
Clusters on Gram stain strongly suggest staphylococci, but MRSA cannot be concluded from shape alone. MRSA requires antibiotic-resistance testing (for example, mec-mediated resistance screening and susceptibility testing), so clinicians still need culture and lab guidance to choose the right drug.
Can staphylococci cause pus but still be missed on the first culture?
Yes. Prior antibiotic use, inadequate specimen collection, or culturing the wrong material can reduce yield. If symptoms persist or worsen, clinicians often repeat sampling and request susceptibility testing rather than relying on an old negative result.
Why does a wound with staphylococcal pus sometimes improve without antibiotics?
For uncomplicated abscesses, incision and drainage removes most of the bacterial burden and low-oxygen cavity environment where bacteria thrive. Antibiotics are then used mainly for systemic signs, rapidly spreading infection, high-risk patients, or difficult-to-drain locations.
What’s the most important practical sign that cellulitis is becoming urgent, not routine?
Rapidly expanding redness along with fever, chills, or systemic symptoms is the key escalation pattern. Another red flag is quickly increasing pain out of proportion to the visible skin changes.
If someone has recurrent boils, should they assume it’s always the same bacteria and treat the same way?
Not necessarily. Recurrent abscesses should usually be cultured first to identify the organism and any resistance pattern. Repeated empiric choices without culture can fail if the pathogen or susceptibility profile has changed.
Do staphylococci in pus mean the patient needs decolonization, and when is that considered?
Decolonization is considered for selected people with recurrent staphylococcal infections, especially if household transmission is suspected. It typically follows clinician assessment and is not a one-size-fits-all step after a single uncomplicated episode.
How should pus drainage be handled at home if MRSA is a possibility?
Treat any draining wound as potentially containing resistant staphylococci: cover with absorbent dressings, use disposable gloves for dressing changes, avoid contact with others’ personal items, and properly bag and dispose of used materials. Afterward, wash hands thoroughly with soap and water.
Does low oxygen in a wound always mean you’ll have biofilm, and should that change treatment?
Low oxygen supports bacterial survival and helps infections persist, but biofilm is not guaranteed. However, if infections are recurrent, device-associated, or not clearing despite drainage and appropriate antibiotics, clinicians often consider biofilm and may pursue additional strategies such as device management or extended/targeted therapy.
For food safety, what’s the practical difference between preventing growth and preventing toxin already made?
Cooling prevents additional bacterial growth, but if toxin-producing levels have already been reached, reheating may not reliably make the food safe. That’s why strict time-and-temperature control within the critical window is emphasized.
Can staphylococcal food contamination come from asymptomatic carriers with no obvious wound?
Yes. People with colonization in the nose or on skin can contaminate food through hand contact during preparation, especially if they have minor skin problems or poor hand hygiene. This is why handwashing compliance matters even when no wound is visible.
Are staphylococci only a concern for external skin infections?
No. Staphylococci also cause infections related to implanted devices and urinary tract infections (for specific species). Device biofilms can make infections stubborn, so persistent symptoms in the context of hardware often require specialized clinical management.
Does Penicillin Mold Grow on Bread? Safety and Storage Tips
Explains penicillin vs mold on bread, why conditions enable growth, how to spot it, and safe storage to prevent it.


