Yes, Micrococcus luteus can grow on mannitol salt agar (MSA), but it will not ferment mannitol. That means you will see visible colonies on the plate, but the medium around them stays pink or red, not yellow. That pink medium is actually the expected, textbook result for Micrococcus luteus on MSA, and it is an important distinction from what you would see with Staphylococcus aureus.
Does Micrococcus luteus grow on mannitol salt agar?
What MSA is actually designed to do
Mannitol salt agar is a selective and differential medium. The two jobs are separate, and understanding both helps you read results correctly.
The selective part comes from 7.5% sodium chloride (75 g/L). That salt concentration is high enough to inhibit most bacteria. Only halotolerant or salt-adapted organisms can survive it, which is why MSA is used to selectively isolate staphylococci from mixed samples. The FDA BAM formulation also includes phenol red (0.025 g/L) and targets a final pH of 7.4, with mannitol as the fermentable carbohydrate.
The differential part comes from phenol red combined with mannitol. If an organism ferments mannitol, it produces acid, drops the pH, and turns the phenol red indicator yellow. If it grows but does not ferment mannitol, the medium stays its original red-pink color. That color shift is the readout you are interpreting, not the colony color itself.
Does Micrococcus luteus grow on MSA?
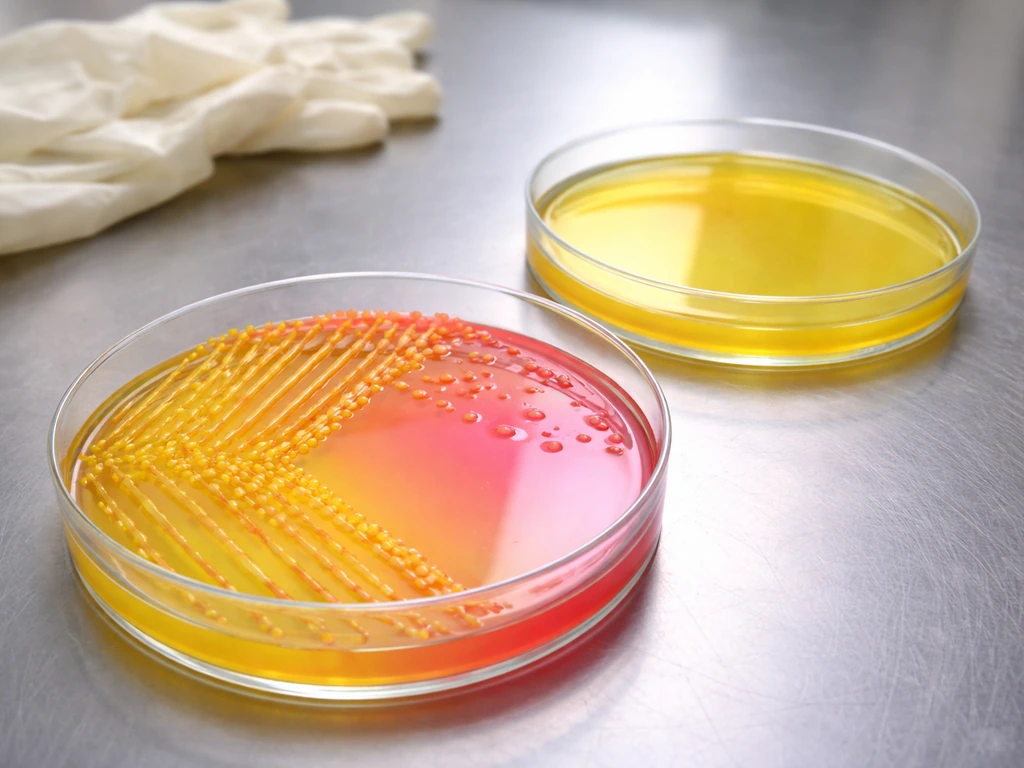
Micrococcus luteus does grow on MSA. Gram-negative bacteria are generally less likely to grow on MSA, but it depends on salt tolerance and other growth requirements. It is salt-tolerant enough to survive the 7.5% NaCl and produce visible colonies. Research on M. luteus K-15 has shown a minimum NaCl requirement around 0.5 M, which puts it comfortably in the halotolerant category, well able to handle MSA's osmotic pressure.
What it does not do is ferment mannitol. The ASM image gallery and multiple manufacturer IFUs confirm this directly: Micrococcus luteus on MSA shows growth with no mannitol fermentation, leaving the medium pink or red-orange. That is your expected result.
Here is where people get tripped up. M. luteus produces naturally yellow-pigmented colonies. So you can have a plate with small, bright yellow colonies sitting in a medium that stays pink. That yellow color on the colony does not mean mannitol fermentation. The medium has to turn yellow for that to count.
How Micrococcus and Staphylococcus look different on MSA
When you put these organisms side by side on MSA, the differences are visible but require careful interpretation. Here is how a typical comparison looks:
| Organism | Growth on MSA | Medium Color Change | Colony Color |
|---|---|---|---|
| Staphylococcus aureus | Yes (good growth) | Yellow (mannitol fermentation) | White to cream/yellow |
| Micrococcus luteus | Yes (moderate growth) | None (stays pink/red) | Bright yellow (natural pigment) |
| Staphylococcus epidermidis | Partial to inhibited | None (stays pink/red) | White to off-white |
| Most gram-negatives | Inhibited/no growth | No change | N/A |
The key takeaway from that table: the medium color change is everything. S. aureus turns the whole medium around the colony yellow because of acid production from mannitol fermentation. M. luteus colonies may look yellow themselves, but if the surrounding medium is still pink, that is a non-fermenter result. The same non-fermenter result applies to S. epidermidis when it does manage to grow, which is relevant if you are comparing MSA behavior across gram-positive cocci. If you are specifically asking whether S. epidermidis grows on MSA, the answer is yes, but you still need to check the medium color change to interpret mannitol fermentation.
Why your results might look different than expected
A few variables can push your MSA results in unexpected directions, so it helps to know what affects the readout.
Salt concentration and strain variation
Not every Micrococcus strain tolerates salt equally well. Most M. luteus strains are halotolerant and will produce measurable colony growth on MSA, but a marginal strain or a stressed culture might produce sparse or delayed growth that is easy to miss at 24 hours. If you see no growth, do not write it off as inhibition until you have checked incubation conditions and extended to 48 hours.
Incubation time and temperature
Standard MSA incubation is aerobic at 35 to 37°C for 36 to 48 hours. Thermo Fisher's IFU specifies 36°C plus or minus 1°C. Reading too early at 24 hours can undercount growth and miss faint color changes. M. luteus grows well at 37°C, so temperature is rarely the issue, but cutting the incubation short can leave you with an ambiguous plate.
Inoculum size
A very heavy inoculum can make even non-fermenters look like they are acidifying the medium, especially if metabolic activity at high cell density shifts the pH slightly. Streak for isolated colonies rather than flooding the plate, and use a fresh, well-grown culture for the most reliable results.
The colony pigment trap
This one deserves its own mention because it catches people regularly. M. luteus produces a natural carotenoid yellow pigment in its colonies. If you glance at the plate and see yellow, it is easy to reflexively record mannitol fermentation. Always look at the medium around and between the colonies, not just the colony color itself. The medium has to shift to yellow for a positive fermentation result.
Running and reading an MSA test correctly

If you are running MSA right now and want to be confident in your results, here is a practical setup that gives you the clearest read:
- Use fresh MSA plates at the correct pH (7.4). Plates that have been stored too long or improperly can have degraded phenol red or absorbed moisture that affects the color baseline.
- Streak for isolation, not mass inoculation. Isolated colonies are essential for reading the local medium color change accurately.
- Include controls on every run: S. aureus as a positive control (yellow medium expected) and S. epidermidis or M. luteus itself as a non-fermentation control (pink medium expected).
- Incubate aerobically at 35 to 37°C. MSA is an aerobic medium and M. luteus is a strict aerobe, so anaerobic or microaerophilic conditions will not give you a valid result.
- Read at 36 to 48 hours. Check at 24 hours as an interim, but do not make final calls until the full incubation window.
- When reading, look at the medium color, not the colony color. Record yellow medium as mannitol fermentation positive, pink or red medium as negative.
After incubation, if you have colonies with a non-fermentation result (pink medium), you need more than MSA to tell you what organism you are looking at. M. luteus growing on MSA looks a lot like other non-fermenting gram-positive cocci at this stage.
MSA alone is not enough to confirm identity
MSA tells you two things: whether an organism can tolerate 7.5% NaCl, and whether it ferments mannitol. It cannot distinguish Micrococcus luteus from coagulase-negative staphylococci or other halotolerant cocci by growth and color change alone. For anyone in a food safety or clinical context, that is a real limitation.
Thermo Fisher's IFU is explicit about this: subculture any suspect colonies from MSA onto a non-selective medium before running confirmatory tests. The high salt environment on MSA can stress cells and interfere with downstream biochemical results if you test directly from the MSA plate.
The most useful confirmatory steps after MSA for distinguishing Micrococcus luteus from staphylococci are catalase and oxidase testing. M. luteus is both catalase-positive and oxidase-positive, and it is a strict aerobe. Most staphylococci are catalase-positive but oxidase-negative, and they are facultative anaerobes. That oxidase difference is one of the clearest biochemical separators between Micrococcus and Staphylococcus. After that, a coagulase test will confirm or rule out S. aureus, which is the organism MSA was primarily designed to find. For more context on what environments Staphylococcus aureus prefers, see where does staphylococcus aureus grow.
Quick confirmatory checklist after MSA
- Subculture from MSA to nutrient agar or blood agar to get clean, unstressed colonies
- Run catalase test: both Micrococcus and Staphylococcus are positive, so this alone does not separate them
- Run oxidase test: Micrococcus luteus is oxidase-positive; Staphylococcus aureus is oxidase-negative
- Run coagulase test: S. aureus is coagulase-positive; M. luteus and coagulase-negative staphylococci are negative
- Check colony morphology and Gram stain: M. luteus forms tetrads or clusters of larger cocci; staphylococci form irregular clusters
- Consider bacitracin susceptibility if lab resources are limited: Micrococcus spp. are typically susceptible; Staphylococcus spp. are resistant
If you are working in a food safety context specifically, the practical concern with MSA is almost always about identifying S. aureus as a potential pathogen. M. luteus growing on MSA without mannitol fermentation is not a safety flag on its own, but it does remind you that MSA growth is not synonymous with S. aureus contamination. Any non-yellow result on MSA still needs follow-up before you can make a call about what organism you actually have.
FAQ
If Micrococcus luteus colonies look yellow on MSA, how do I tell whether mannitol fermentation actually happened?
Ignore colony color alone and focus on the phenol red indicator in the agar around and between colonies. A true positive for mannitol fermentation gives a yellow color change in the medium, not just yellow pigmentation in the cells.
What incubation issue could cause me to incorrectly conclude Micrococcus luteus did not grow on MSA?
Reading too early. If you check at 24 hours, you may miss sparse or delayed halotolerant growth. Incubate within the stated window (often up to 48 hours) and re-read for faint pink-to-yellow shifts.
Can a heavy inoculum make the MSA result misleading for Micrococcus luteus?
Yes. Very dense inoculation can shift pH slightly through overall metabolism, producing a confusing color change even when mannitol fermentation is weak or absent. Use isolated streaks and a well-grown culture to keep interpretation clean.
Does “no yellow medium” mean Micrococcus luteus is definitely present if I see growth on MSA?
No. MSA can show halotolerant growth without mannitol fermentation for several gram-positive cocci. You still need confirmatory tests (for example, oxidase, catalase, and then coagulase if staphylococcal identity is suspected).
If Micrococcus luteus grows on MSA but does not ferment mannitol, could it still turn the medium yellow in some situations?
It can be uncommon, but borderline or stressed cultures may produce ambiguous, partial, or delayed color changes. Treat any equivocal yellowing cautiously and repeat with fresh isolated colonies and appropriate incubation before concluding fermentation.
Do I need to subculture off MSA before doing biochemical confirmation tests?
It is strongly recommended. The high-salt conditions and any residual nutrients on MSA can stress cells or interfere with downstream biochemical reactions. Subculture suspect colonies onto a non-selective medium first, then test.
What confirmatory results most help distinguish Micrococcus luteus from staphylococci after MSA?
Oxidase and catalase are especially useful here. Micrococcus luteus is typically catalase-positive and oxidase-positive (and is a strict aerobe), while many staphylococci are oxidase-negative.
If I specifically want to know whether Micrococcus luteus is salt-tolerant enough for MSA, what practical readout should I use?
Use the fact of growth on the 7.5% NaCl selective system as the practical indicator, but be aware that “growth vs no growth” can depend on culture health and timing. Confirm with a second plate or extended incubation if growth is minimal or delayed.
Can Bacteria Grow in Vinegar? pH, Survival, and Safety Tips
Explore if bacteria can grow in vinegar, how pH and dilution affect survival, and practical food-safety storage tips.


