Chocolate agar is specifically designed to grow fastidious bacteria that cannot survive on standard media. The organisms most reliably recovered on it are Haemophilus species (especially Haemophilus influenzae) and Neisseria species (including Neisseria meningitidis and Neisseria gonorrhoeae). A range of other fastidious respiratory and pathogenic bacteria can also grow, but the medium is not a catch-all. Most common environmental and food-spoilage organisms will grow too, but the real reason anyone reaches for chocolate agar is to recover those demanding, fastidious pathogens that nothing else supports as well.
What Bacteria Grow on Chocolate Agar and Why

What chocolate agar actually is and why it works
Chocolate agar (often abbreviated CHOC) starts as standard blood agar, but the red blood cells are gently heated until they lyse. That heating turns the agar from red to a warm brown color, which is where the name comes from. The key effect of lysis is that intracellular contents are released directly into the medium. Most importantly, hemin (factor X) and NAD+ (factor V) become freely available in the agar. These are the two growth factors that fastidious bacteria like Haemophilus influenzae require and cannot make on their own.
On regular blood agar, hemin stays bound inside intact red blood cells. Unless something lyses those cells, factor X is not accessible. That is exactly why H. influenzae cannot grow properly on blood agar but thrives on chocolate agar. Because H. influenzae needs growth factors that are released by the heat-lysing step, it does not grow properly on blood agar H. influenzae grow on blood agar. The heat-lysis step is the entire functional difference between the two media, and it is the reason chocolate agar exists as a separate medium rather than just being a redundant version of blood agar.
Chocolate agar is nonselective, meaning it does not contain antibiotics or other inhibitory agents by default. If you want selectivity, you need a variant like Thayer-Martin medium, which adds antibiotics to suppress commensal flora. Standard chocolate agar grows whatever is viable and compatible, which is useful for sterile-site specimens but less useful when the sample is loaded with competing organisms.
Which bacteria actually grow on chocolate agar

The organisms chocolate agar is genuinely built for fall into a few well-defined groups. Here is a practical breakdown:
Haemophilus species
Haemophilus influenzae is the textbook organism for chocolate agar. It requires both factor X (hemin) and factor V (NAD+), neither of which it can synthesize itself. Because chocolate agar supplies both factors freely, H. influenzae grows reliably on it and poorly or not at all on blood agar alone. Colonies appear small, around 0.5 to 1 mm, smooth, translucent, and convex with an entire edge. Other Haemophilus species may require only one of the two factors. H. parainfluenzae, for example, needs only factor V. These species-level differences in factor requirements are precisely what X/V factor testing is designed to sort out after you have grown the organism.
Neisseria species

Neisseria meningitidis and Neisseria gonorrhoeae both grow on chocolate agar, though they need supplemental CO2 to do so reliably (more on that below). Because Neisseria meningitidis and Neisseria gonorrhoeae can grow on chocolate agar, you can use it to recover these fastidious organisms when the incubation conditions are correct. N. meningitidis produces large, colorless to gray opaque colonies. N. gonorrhoeae colonies tend to be smaller, grayish-white to colorless, and often appear mucoid. Neither grows well on standard chocolate agar without CO2 enrichment, which is a common point of failure in labs that do not adjust their incubator atmosphere.
Other fastidious organisms
Beyond Haemophilus and Neisseria, chocolate agar supports a broader group of fastidious organisms, including HACEK group bacteria (Haemophilus, Aggregatibacter, Cardiobacterium, Eikenella, and Kingella species), Moraxella catarrhalis, some Brucella species, and Pasteurella species. Many of these are respiratory or mucosal pathogens that share the fastidious growth requirement that makes chocolate agar the right medium to reach for.
It is worth noting that non-fastidious organisms like Staphylococcus aureus, Streptococcus pneumoniae, and members of the Enterobacteriaceae family will also grow on chocolate agar because the medium does not inhibit them. But the medium is not designed with those organisms in mind, and you would typically use blood agar, MacConkey agar, or other media to characterize them. Chocolate agar is chosen specifically when you need to recover organisms that cannot grow without supplemental growth factors.
| Organism | Factor X Required | Factor V Required | Grows on Blood Agar | Grows on Chocolate Agar |
|---|---|---|---|---|
| H. influenzae | Yes | Yes | No (satellite only) | Yes |
| H. parainfluenzae | No | Yes | Weakly/variable | Yes |
| N. meningitidis | No | No | Yes (with CO2) | Yes (with CO2) |
| N. gonorrhoeae | No | No | Yes (with CO2) | Yes (with CO2) |
| S. aureus | No | No | Yes | Yes |
| HACEK group | Variable | Variable | Poorly/rarely | Yes |
How incubation conditions shape what you actually see

Getting chocolate agar right is not just about the medium. Incubation conditions determine whether fastidious organisms grow at all, and choosing the wrong settings is a reliable way to get a false-negative result.
Temperature
The standard incubation temperature for fastidious respiratory pathogens on chocolate agar is 35 to 37°C. The CDC recommends 35°C to 36.5°C specifically for N. gonorrhoeae. UK Standards for Microbiology Investigations specify 35 to 37°C for Haemophilus species. Going outside that narrow window matters. Temperatures below 35°C slow or prevent growth of these organisms, and temperatures above 37.5°C can also reduce recovery.
CO2 atmosphere
Supplemental CO2 at 5 to 10% is not optional for Neisseria species and is strongly beneficial for Haemophilus. Some Neisseria sources state flatly that growth on chocolate agar does not occur without added CO2. A candle extinction jar (which generates roughly 3 to 5% CO2) is a minimal workaround, but a calibrated CO2 incubator set to 5% is standard practice. If you are incubating in ambient air and wondering why nothing grew, CO2 is almost always the first thing to check.
Incubation time
Most clinically relevant fastidious organisms show visible growth within 18 to 24 hours under proper conditions. Chocolate agar plates are typically examined at 24 hours and again at 48 hours before being reported as negative. Haemophilus colonies may be visible at 18 hours but are more reliably identified at 24 hours. Neisseria gonorrhoeae is typically recovered within 18 to 24 hours. Extending incubation to 48 hours catches slower-growing or lower-inoculum samples, especially from sterile-site specimens.
Recognizing growth and telling real growth from background noise
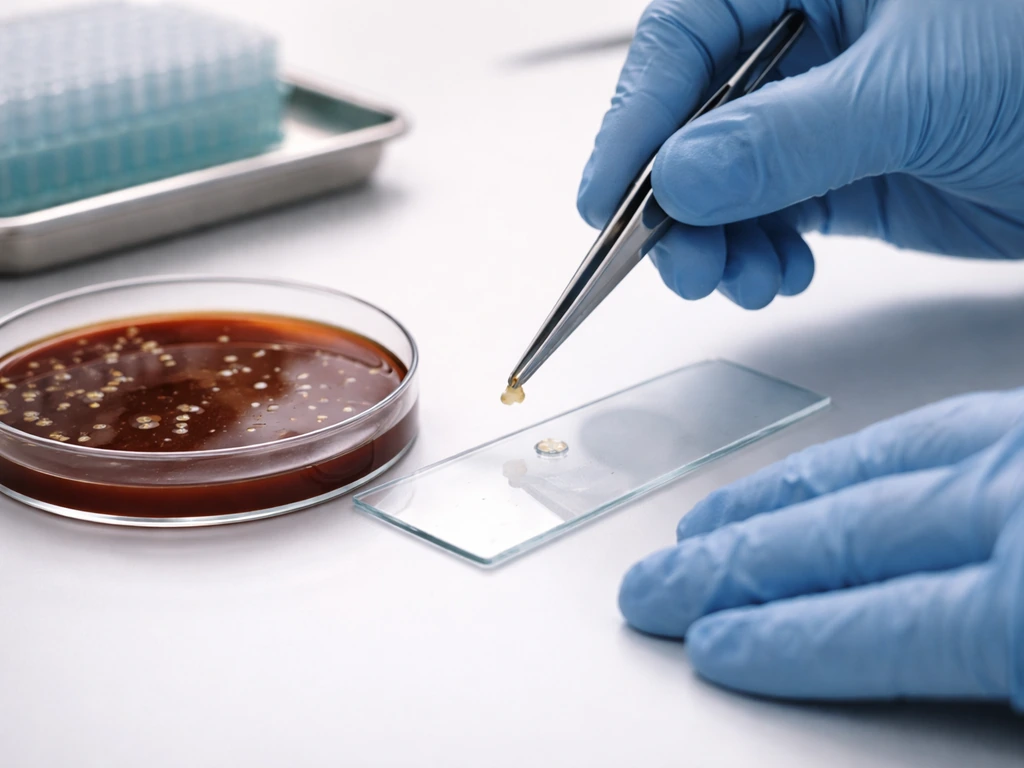
On a properly incubated chocolate agar plate, true growth from a fastidious organism looks distinct once you know what to expect. H. influenzae colonies are small (0.5 to 1 mm), smooth, translucent, and slightly convex. They often have a faint mousy or bleach-like odor. Neisseria meningitidis produces larger colonies, colorless to gray-opaque. Neisseria gonorrhoeae tends to be smaller, grayish-white, and mucoid.
The main confusion comes from mixed-culture plates where commensal or contaminant organisms outgrow the target. If your specimen is from a non-sterile site, competing flora can crowd out fastidious organisms or make colonies harder to spot. In that situation, a selective variant of chocolate agar (like Thayer-Martin or Martin-Lewis for Neisseria) would have been the better choice at the outset.
A practical check: if you see no growth at 24 hours, verify your incubation conditions before calling the plate negative. Confirm the incubator was at 35 to 37°C with 5% CO2, and check that the medium was not expired or improperly stored. Use a positive control strain (H. influenzae ATCC 10211 or N. meningitidis ATCC 13090 are standard QC organisms for chocolate agar) so you have something to compare against. If the control grew and the clinical plate did not, the medium is working and you can be confident in a negative result.
What to do after you see growth: confirming what actually grew
Growth on chocolate agar alone tells you an organism is viable and compatible with the medium. It does not tell you what it is. These are the standard follow-up steps used to get to a species-level identification.
- Gram stain first: Haemophilus species are small Gram-negative coccobacilli. Neisseria species are Gram-negative diplococci (kidney-bean shaped pairs). This narrows the field quickly and tells you which confirmatory tests to run next.
- Oxidase test: Haemophilus influenzae is oxidase-positive. Neisseria species are also oxidase-positive. A positive oxidase result from a Gram-negative organism on chocolate agar strongly points toward either group.
- Catalase test: H. influenzae is catalase-positive. This is quick and adds another data point alongside oxidase.
- X and V factor testing: Disk or strip tests with hemin (X), NAD+ (V), and XV combined disks on a factor-free medium like tryptic soy agar confirm which growth factors the organism requires. This is the key step for identifying and differentiating Haemophilus species from each other.
- Satellitism test on blood agar: Streak S. aureus across a blood agar plate, then streak your isolate perpendicular to it. Haemophilus species that need factor V will grow as satellite colonies close to the S. aureus streak, where NAD+ is released by the staphylococci. This is a classic confirmatory observation.
- MALDI-TOF MS: In modern clinical labs, colonies grown on chocolate agar at 37°C in 5% CO2 overnight are routinely processed directly by MALDI-TOF for rapid species identification. It is faster than biochemical panels and handles Haemophilus and Neisseria well.
- PCR or molecular testing: If culture failed but infection is still suspected (for example, after antibiotic treatment has already begun), PCR can detect Haemophilus influenzae and Neisseria meningitidis DNA even from nonviable organisms. Culture alone can miss organisms in treated patients.
For Neisseria gonorrhoeae specifically, the CDC's presumptive identification criteria involve finding Gram-negative, oxidase-positive diplococci from an appropriate specimen cultured under the correct conditions. Confirmatory steps include subculture onto fresh nonselective chocolate agar for a pure colony, followed by carbohydrate utilization tests, MALDI-TOF, or nucleic acid amplification testing depending on your lab's workflow.
One practical note on specimen source: the CDC recommends using nonselective chocolate agar for sterile-site specimens (like CSF or blood) and selective variants for non-sterile sites like urogenital swabs. If you are trying to recover Neisseria gonorrhoeae from a urogenital specimen on plain chocolate agar, you may be making recovery harder than it needs to be by allowing commensal organisms to compete.
The sibling questions of whether specific organisms like H. influenzae, N. gonorrhoeae, or S. aureus grow on chocolate agar each have their own nuances in terms of factor requirements and incubation conditions, but the underlying logic is always the same: match the medium and conditions to what the target organism needs, then confirm identity with biochemical or molecular follow-up rather than relying on growth alone as identification.
FAQ
If chocolate agar is nonselective, how can I tell whether the plate actually supported the intended fastidious organism versus just “something” growing?
Check for organisms with expected morphology for the target, then verify with a control strain processed the same day. In practice, if you have heavy mixed flora, true fastidious growth can be missed, so you often need to subculture suspected colonies onto fresh chocolate agar under the correct CO2 conditions for confirmation.
Does a fastidious organism grow on chocolate agar if the plate is incubated in ambient air?
Some can show limited growth, but Neisseria species are the most sensitive, and lack of CO2 commonly leads to false negatives. If you incubated without CO2, treat a negative result as inconclusive for Neisseria and repeat with 5% CO2 (or an equivalent validated system) when clinically appropriate.
What should I do if Haemophilus or Neisseria growth is weak or delayed compared with the expected 18 to 24 hour window?
First confirm incubator calibration and gas concentration, then extend incubation to 48 hours before calling negative (especially for low inoculum or sterile-site specimens). Also review specimen handling, because drying, delayed transport, or prior antibiotics can reduce recovery even when the medium and CO2 are correct.
Can chocolate agar be used to recover Neisseria gonorrhoeae, and what are the common reasons it still fails?
It can, but success depends heavily on CO2 enrichment and appropriate temperature, plus having a viable organism from the specimen. Common failure points are using the wrong incubator atmosphere, letting swabs dry, delayed plating, or not subculturing a presumptive colony onto fresh chocolate agar for purity before confirmatory testing.
Will Staphylococcus aureus or other common bacteria grow on chocolate agar, and does that matter for interpretation?
Yes, many non-fastidious organisms grow because the medium does not include inhibitory agents. The practical issue is interpretive, mixed growth can mask target colonies, so you should not rely on “growth occurred” as evidence of fastidious organisms, and you may need selective media for non-sterile specimens.
How do I decide between plain chocolate agar and a selective variant like Thayer-Martin for a given specimen?
Use plain chocolate agar when the specimen is from a sterile site (lower competing flora), and use selective variants for non-sterile sites such as urogenital swabs to suppress commensals. If you suspect Neisseria from a urogenital specimen, starting with a selective Neisseria-focused medium generally improves recovery.
If nothing grows on chocolate agar at 24 hours, can I report it as negative immediately?
Not usually for fastidious targets. The common approach is to examine at 24 hours and again at 48 hours, because slower-growing strains, low inoculum, or suboptimal specimen quality can produce late or scant colonies even when conditions are otherwise acceptable.
What positive control should I use, and how does it help beyond confirming the medium is “not bad”?
Include a known QC strain for the target organism (such as a standard Haemophilus or Neisseria QC) and incubate under the same atmosphere and temperature as the clinical plates. If the control grows but the clinical plate does not, the medium and incubation are likely fine, shifting attention to specimen viability, inoculum, or antibiotic exposure.
How long can chocolate agar plates be stored before use, and does age affect growth results?
Expired or improperly stored plates can reduce recovery, especially for fastidious organisms that rely on intact released factors and consistent moisture. If you suspect poor performance, compare with a QC run and check storage conditions such as temperature, light exposure, and time since preparation, then retest if needed.
Does “growth on chocolate agar” tell me the exact species or just that it’s viable?
It only tells you the organism can grow under those conditions. Species identification requires additional testing, typically colony purification followed by biochemical tests, MALDI-TOF, or nucleic acid methods, depending on your workflow.
Does Haemophilus influenzae Grow on Blood Agar? Guide
Does Haemophilus influenzae grow on blood agar? Compare H. influenzae vs H. parainfluenzae and when to expect growth or


